Chemistry, 01.07.2020 15:01, bluetigerbird4745
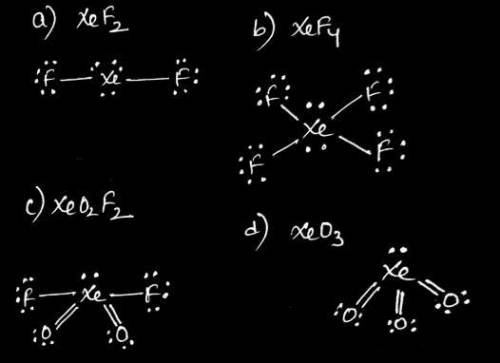
Of the noble gases, only Kr, Xe, and Rn are known to form a few compounds with O and/or F. Complete the following Lewis structures by adding all of the necessary lone pairs and any multiple bonds for each molecule below. Draw your structure with the lowest possible formal charges.
A) XeF2
B) XeF4
C) XeO2F2
D) XeO3

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 11:50, trinityrae4657
Acompound has a molecular weight of 12.124 atomic mass units and the empirical formula c3h40. what is the molecular formula of the compound?
Answers: 3

Chemistry, 22.06.2019 15:30, lovebaeforlife351
Each of the following reactions is allowed to come to equilibrium and then the volume is changed as indicated. predict the effect (shift right, shift left, or no effect) of the indicated volume change. drag the appropriate items to their respective bins. co(g) + h2o(g) < => co2(g) + h2(g) (volume is decreased) pcl3(g) + cl2(g) < => pcl5(g) (volume is increased) caco3(s)< => cao(s) + co2(g) (volume is increased)
Answers: 1

Do you know the correct answer?
Of the noble gases, only Kr, Xe, and Rn are known to form a few compounds with O and/or F. Complete...
Questions in other subjects:






Social Studies, 06.01.2020 23:31

Chemistry, 06.01.2020 23:31