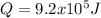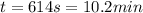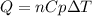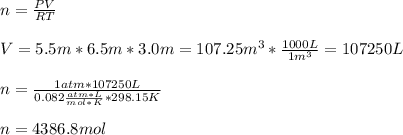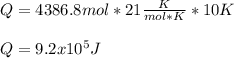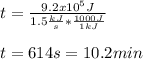The heat capacity of air is much smaller than that of water, and relatively modest amounts of heat are needed to change its temperature. This is one of the reasons why desert region, although very hot during the day, are bitterly cold at night. The heat capacity of air at room temperature and pressure is appoximately 21 J/K*mol. How much energy is required to raise the temperature of a room of dimensions 5.5m x 6.5m x 3.0m by 10 degrees Celsius? If losses are neglected, how long will it take a heater rated at 1.5 kW to achieve that increase given that 1 W = 1 J/s?

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 15:00, asims13
Which element in the third period would you expect to have the larger atomic radius, sodium (na) or sulfur (s)? a. sodium, because it has a higher effective nuclear charge attracting electrons in fewer energy levels. b. sodium, because it has fewer protons attracting electrons in the same energy levels. c. sulfur, because it has more protons attracting electrons in more energy levels. d. sulfur, because it has a higher effective nuclear charge attracting electrons in the same energy levels.
Answers: 2


Chemistry, 22.06.2019 10:40, justicejesusfreak
Which buffer would be better able to hold a steady ph on the addition of strong acid, buffer 1 or buffer 2? explain. buffer 1: a solution containing 0.10 m nh4cl and 1 m nh3. buffer 2: a solution containing 1 m nh4cl and 0.10 m nh3
Answers: 1
Do you know the correct answer?
The heat capacity of air is much smaller than that of water, and relatively modest amounts of heat a...
Questions in other subjects:



Mathematics, 12.11.2019 01:31

Mathematics, 12.11.2019 01:31



Social Studies, 12.11.2019 01:31



Mathematics, 12.11.2019 01:31