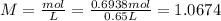Chemistry, 26.06.2020 17:01, villafana36
A 650.0 mL solution contains 125 grams of glucose (C6H12O6). If the molar mass of C6H12O6 is 180.16 g/mol, what is the molarity of this solution? answer options are 0.0106 M C6H12O6 0.0195 M C6H12O6 1.07 M C6H12O6 1.92 M C6H12O6
need help ASAP
will mark brainlest

Answers: 1
Other questions on the subject: Chemistry



Chemistry, 22.06.2019 19:10, krisandlance
Astudent completes a titration by adding 12.0 milliliters of naoh(aq) of unknown concentration to 16.0 milliliters of 0.15 m hcl(aq). what is the molar concentration of the naoh(aq)? 1)5.0 m 2)0.20 m 3)0.11 m 4)1.1 m
Answers: 1

Chemistry, 22.06.2019 20:00, denaemarie02
Nitrogen dioxide decomposes according to the reaction 2 no2(g) ⇌ 2 no(g) + o2(g) where kp = 4.48 × 10−13 at a certain temperature. if 0.70 atm of no2 is added to a container and allowed to come to equilibrium, what are the equilibrium partial pressures of no(g) and o2(g)
Answers: 2
Do you know the correct answer?
A 650.0 mL solution contains 125 grams of glucose (C6H12O6). If the molar mass of C6H12O6 is 180.16...
Questions in other subjects:



English, 27.11.2020 08:10

Mathematics, 27.11.2020 08:10

Advanced Placement (AP), 27.11.2020 08:10


English, 27.11.2020 08:10



Mathematics, 27.11.2020 08:10


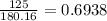 mol de glucosa
mol de glucosa