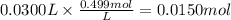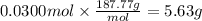Chemistry, 27.06.2020 05:01, viktoria1198zz
Aqueous solutions of copper (II) bromide and silver (1) acetate react to form solid
silver (1) bromide and aqueous copper (II) acetate according to the UNBALANCED
reaction below.
CuBr2 (aq) + AGCH3CO2 (aq)
-
AgBr (s) + Cu(CH3CO2)2 (aq)
How many grams of silver (1) bromide will form if 30.0 mL of 0.499 M copper (II)
bromide react with excess silver (1) acetate?

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 00:00, 20jessicacabriales
The pressure in a fluid is affected by which characteristics of that fluid
Answers: 1

Chemistry, 22.06.2019 10:00, Cythina2007
The reactions shown here can be combined to make the overall reaction c(s) + h2o(g) ⇌ co(g) + h2(g) by reversing some and/or dividing all the coefficients by a number. a. c(s) + o2(g) → co2(g) k=1.363×10^69 b. 2 h2(g) + o2(g) → 2 h2o(g) k=1.389×10^80 c. 2co(g) + o2 (g) → 2 co2(g) k=1.477×10^90
Answers: 1

Chemistry, 22.06.2019 20:00, ahnorthcutt4965
Acm ruler with main graduations from 1 to 10 from left to right there are 10 secondary graduations between each of the main graduations there is a line that begins. at the left end of the ruler 10 secondary graduations to the left of the “1 main graduation the right end of the line ends on the eighth secondary graduation to the right of 3 how long is the line
Answers: 1

Chemistry, 22.06.2019 21:00, taylorlanehart
Use the measurements in the table to determine which unidentified metal has the highest density. metal volume mass a 10.5 cm3 122 g b 14.2 cm3 132 g c 16.1 cm3 115 g d 12.7 cm3 126 g
Answers: 2
Do you know the correct answer?
Aqueous solutions of copper (II) bromide and silver (1) acetate react to form solid
silver (1) brom...
Questions in other subjects:


History, 23.09.2019 18:30




Social Studies, 23.09.2019 18:30

Chemistry, 23.09.2019 18:30

Mathematics, 23.09.2019 18:30