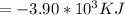Chemistry, 26.06.2020 15:01, coltonduggan
When 1.550 gg of liquid hexane (C6H14)(C6H14) undergoes combustion in a bomb calorimeter, the temperature rises from 25.87 ∘C∘C to 38.13 ∘C∘C. Find ΔErxnΔErxn for the reaction in kJ/molkJ/mol hexane. The heat capacity of the bomb calorimeter, determined in a separate experiment, is 5.73 kJ/∘CkJ/∘C.

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 02:00, lwattsstudent
In the following redox reaction which is the oxidizing agent and which is the reducing agent? alcl3 + na nacl + al
Answers: 1

Chemistry, 22.06.2019 11:00, jodonw5616
The diagram below shows the different phase transitions that occur in matter. which arrow represents the transition in which dew is formed?
Answers: 1


Chemistry, 22.06.2019 12:30, MrSavannahCat
Clyde and marilyn are riding a roller coaster. during which section(s) of the track is their potential energy converted to kinetic energy? a. from point b to point c only b. from point b to point d only c. from point a to point b only d. from point a to point b and from point c to point d
Answers: 1
Do you know the correct answer?
When 1.550 gg of liquid hexane (C6H14)(C6H14) undergoes combustion in a bomb calorimeter, the temper...
Questions in other subjects:


Mathematics, 12.11.2019 19:31

Mathematics, 12.11.2019 19:31


Mathematics, 12.11.2019 19:31


Advanced Placement (AP), 12.11.2019 19:31


History, 12.11.2019 19:31

Social Studies, 12.11.2019 19:31