

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 04:00, lucasrandall
Asolution contains 225 g of sodium chloride, nacl, dissolved in enough water to make a 0.25 l of solution. what is the molarity of the solution?
Answers: 2


Chemistry, 22.06.2019 04:30, logan12345677885675
What are the primary responsibilities of a chemical engineer involved in "r& d"? develop large scale manufacturing operations discover new products and processes training of new chemists determine products needed by consumers
Answers: 2
Do you know the correct answer?
9. Given the following reaction:CO (g) + 2 H2(g) CH3OH (g)In an experiment, 0.45 mol of CO and 0.57...
Questions in other subjects:

Mathematics, 02.09.2019 06:30

Mathematics, 02.09.2019 06:30




English, 02.09.2019 06:30


Physics, 02.09.2019 06:30


English, 02.09.2019 06:30


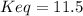

![Keq=\frac{[CH_3OH]}{[CO][H_2]^2}](/tpl/images/0694/1862/81c3a.png)
 due to the reaction extent we can write:
due to the reaction extent we can write:![Keq=\frac{x}{([CO]_0-x)([H_2]_0-2x)^2}](/tpl/images/0694/1862/e4583.png)
![[CO]_0=\frac{0.45mol}{1.00L}=0.45M\\](/tpl/images/0694/1862/3035d.png)
![[H_2]_0=\frac{0.57mol}{1.00L}=0.57M\\](/tpl/images/0694/1862/dc61d.png)
![[CO]_{eq}=\frac{0.28mol}{1.00L}=0.28M\\](/tpl/images/0694/1862/ecf3a.png)
![x=[CO]_0-[CO]_{eq}=0.45M-0.28M=0.17M](/tpl/images/0694/1862/8aaf6.png)





