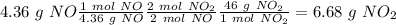Nitrogen monoxide is produced by combustion in an automobile engine. For the following reaction, 4.36 grams of nitrogen monoxide are mixed with excess oxygen gas . The reaction yields 5.46 grams of nitrogen dioxide . nitrogen monoxide ( g ) oxygen ( g ) nitrogen dioxide ( g ) What is the theoretical yield of nitrogen dioxide

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 10:30, kdenormandie3122
Geothermal energy for industrial use is available almost anywhere. a. true b. false
Answers: 2


Chemistry, 22.06.2019 11:40, jerrysandoval22
Which of these expressions are correct variations of the combined gas law? p1v1t2 = p2v2t1 both
Answers: 2
Do you know the correct answer?
Nitrogen monoxide is produced by combustion in an automobile engine. For the following reaction, 4.3...
Questions in other subjects:


Mathematics, 22.04.2021 16:20

Mathematics, 22.04.2021 16:20

Mathematics, 22.04.2021 16:20


Mathematics, 22.04.2021 16:20


English, 22.04.2021 16:20

Mathematics, 22.04.2021 16:20

English, 22.04.2021 16:20


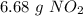
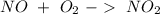
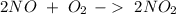
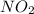 . To do this, we have to first convert the 4.36 g of
. To do this, we have to first convert the 4.36 g of 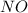 to moles
to moles