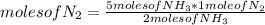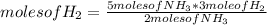N2(g) + 3H2(g) →2NH3(g)
At the end of the chemical reaction, 5 moles of NH3 are produced.
How...

Chemistry, 21.06.2020 22:57, Goldenstate32
N2(g) + 3H2(g) →2NH3(g)
At the end of the chemical reaction, 5 moles of NH3 are produced.
How many moles of N2 and H2 entered the reaction?

Answers: 1
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 03:40, 19thomasar
Kc = 0.040 for the system below at 450oc. if a reaction is initiated with 0.40 mole of cl2 and 0.40 mole of pcl3 in a 2.0 liter container, what is the equilibrium concentration of cl2 in the same system? pcl5(g) ⇄ pcl3(g) + cl2(g)
Answers: 3

Chemistry, 22.06.2019 09:30, matpakootas521
Why do cells appear different in distilled water than they do in 10% salt water?
Answers: 2

Chemistry, 22.06.2019 10:10, ragegamer334p3xlso
Stage in which a star’s outer layers have started to cool and grow outward?
Answers: 3
Do you know the correct answer?
Questions in other subjects:


Arts, 02.03.2021 01:00

English, 02.03.2021 01:00

English, 02.03.2021 01:00


Health, 02.03.2021 01:00

Mathematics, 02.03.2021 01:00


Arts, 02.03.2021 01:00

Geography, 02.03.2021 01:00