
Chemistry, 19.06.2020 10:57, kcombest7219
Amylose is a "polysaccharide" that plants use to store energy. It is made of repeating subunits of C6H10O5. If a particular amylose molecule has 2537 of these subunits, what is its molecular formula? What is its molar mass? What is the empirical formula of amylose?

Answers: 3
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 05:30, medlinalex
Compare and contrast physical changes with chemical changes.
Answers: 1

Chemistry, 22.06.2019 12:30, Svetakotok
Suppose you wanted to make 100 grams of water. what is the molar mass of water (h2o)?
Answers: 2

Chemistry, 22.06.2019 13:30, kassandrarosario1115
How many protons, electrons, and neutrons are in each of the following isotopes? a. zirconium-90 b. palladium-108 c. bromine-81 d. antimony-123
Answers: 1
Do you know the correct answer?
Amylose is a "polysaccharide" that plants use to store energy. It is made of repeating subunits of C...
Questions in other subjects:

Mathematics, 13.11.2020 14:00

Chemistry, 13.11.2020 14:00

English, 13.11.2020 14:00


Social Studies, 13.11.2020 14:00



Biology, 13.11.2020 14:00

Biology, 13.11.2020 14:00

Mathematics, 13.11.2020 14:00


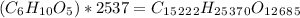
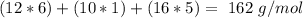

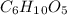 .
.



