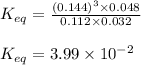Chemistry, 19.06.2020 03:57, KillerSteamcar
Steam reforming of methane (CH4 ) produces "synthesis gas," a mixture of carbon monoxide gas and hydrogen gas, which is the starting point for many important industrial chemical syntheses. An industrial chemist studying this reaction fills a tank with of methane gas and of water vapor, and when the mixture has come to equilibrium measures the amount of carbon monoxide gas to be 18 mol . Calculate the concentration equilibrium constant for the steam reforming of methane at the final temperature of the mixture. Round your answer to significant digits.

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 13:30, justinerodriguz2878
What are the major types of a chemical compound
Answers: 2

Chemistry, 22.06.2019 21:00, lucyamine0
As we move from left to right across the periodic table, what is the general trend? a) atomic radii increase. b) electronegavitiy decreases. c) nuclear shielding increases. d) metallic character decreases.
Answers: 1


Chemistry, 23.06.2019 05:40, shelbylynn1093
Why is it incorrect to balance a chemical equation by changing the subscripts? explain.
Answers: 2
Do you know the correct answer?
Steam reforming of methane (CH4 ) produces "synthesis gas," a mixture of carbon monoxide gas and hyd...
Questions in other subjects:

Mathematics, 01.08.2019 17:30


Mathematics, 01.08.2019 17:30

Mathematics, 01.08.2019 17:30



Mathematics, 01.08.2019 17:30

Health, 01.08.2019 17:30


Mathematics, 01.08.2019 17:30


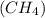 produces "synthesis gas," a mixture of carbon monoxide gas and hydrogen gas, which is the starting point for many important industrial chemical syntheses. An industrial chemist studying this reaction fills a 125 L tank with 20 mol of methane gas and 10 mol of water vapor at 38 degrees celsius. He then raises the temperature, and when the mixture has come to equilibrium measures the amount of hydrogen gas to be 18 mol . Calculate the concentration equilibrium constant for the steam reforming of methane at the final temperature of the mixture. Round your answer to significant digits.
produces "synthesis gas," a mixture of carbon monoxide gas and hydrogen gas, which is the starting point for many important industrial chemical syntheses. An industrial chemist studying this reaction fills a 125 L tank with 20 mol of methane gas and 10 mol of water vapor at 38 degrees celsius. He then raises the temperature, and when the mixture has come to equilibrium measures the amount of hydrogen gas to be 18 mol . Calculate the concentration equilibrium constant for the steam reforming of methane at the final temperature of the mixture. Round your answer to significant digits.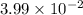
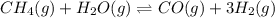
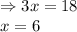
![K_{eq}=\frac{[H_2]^3[CO]}{[CH_4][H_2O]}](/tpl/images/0689/6394/52809.png)
![[H_2]=\frac{18}{125}=0.144M](/tpl/images/0689/6394/7d094.png)
![[CO]=\frac{6}{125}=0.048M](/tpl/images/0689/6394/f476f.png)
![[CH_4]=\frac{14}{125}=0.112M](/tpl/images/0689/6394/df9f1.png)
![[H_2O]=\frac{4}{125}=0.032M](/tpl/images/0689/6394/b4425.png)