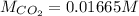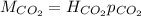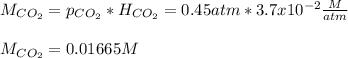Chemistry, 13.06.2020 21:57, destinybowers18
The partial pressure of CO2 gas above the liquid in a carbonated drink is 0.45 atm. Assuming that the Henry's law constant for CO2 in the drink is that same as that in water, 3.7 x 10-2 mol/L atm, calculate the solubility of carbon dioxide in this drink.

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 10:00, JOEFRESH10
Suppose the universe were completely empty except for one object-a solid sphere moving through space of 100 km/s. what sort of path would the object be moving in? explain your answer
Answers: 1



Chemistry, 22.06.2019 18:00, rodriguezscarlet1713
Hydrogenation reactions, in which h2 and an "unsaturated" organic compound combine, are used in the food, fuel, and polymer industries. in the simplest case, ethene (c2h4) and h2 form ethane (c2h6). if 140 kj is given off per mole of c2h4 reacting, how much heat (in mj) is released when 12 kg of c2h6 forms?
Answers: 2
Do you know the correct answer?
The partial pressure of CO2 gas above the liquid in a carbonated drink is 0.45 atm. Assuming that th...
Questions in other subjects:

Mathematics, 29.04.2021 01:00



Mathematics, 29.04.2021 01:00


Computers and Technology, 29.04.2021 01:00

Mathematics, 29.04.2021 01:00



Mathematics, 29.04.2021 01:00