
Chemistry, 13.06.2020 09:57, hmskevinjacobo5471
I NEED HELP PLEASE, THANKS!
Ammonia (NH3) is an example of a Brønsted-Lowry Base.
-Define the Brønsted-Lowry acid-base theory.
-What is the pH of an ammonia solution that has a concentration of 0.335 M? The Kb of ammonia is 1.8 × 10^–5.

Answers: 3
Other questions on the subject: Chemistry



Chemistry, 22.06.2019 21:00, melissalopez12
Acandle’s wick is the fabric string that holds the flame, and it burns down at a constant slow pace when the candle is lit. the wick is usually surrounded by wax. which is the most important property of covalent compounds that makes them useful for making candle wax? a low boiling point a low melting point a high boiling point a high melting point
Answers: 1

Chemistry, 22.06.2019 22:30, needhelpasap8957
Why is the bottom layer of a trophic pyrimid the
Answers: 2
Do you know the correct answer?
I NEED HELP PLEASE, THANKS!
Ammonia (NH3) is an example of a Brønsted-Lowry Base.
-Define the...
-Define the...
Questions in other subjects:

Chemistry, 08.06.2021 01:00


Mathematics, 08.06.2021 01:00

Business, 08.06.2021 01:00

Mathematics, 08.06.2021 01:00

Mathematics, 08.06.2021 01:00

English, 08.06.2021 01:00


Mathematics, 08.06.2021 01:00


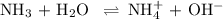
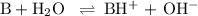
![\rm K_{\text{b}} = \dfrac{\text{[BH}^{+}]\text{[OH}^{-}]}{\text{[B]}} = 1.8 \times 10^{-5}\\\\\dfrac{x^{2}}{0.335 - x} = 1.8 \times 10^{-5}](/tpl/images/0685/0322/975fe.png)
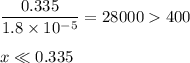
![\dfrac{x^{2}}{0.335} = 1.8 \times 10^{-5}\\\\x^{2} = 0.335 \times 1.8 \times 10^{-5}\\x^{2} = 6.03 \times 10^{-6}\\x = \sqrt{6.03 \times 10^{-6}}\\x = \text{[OH]}^{-} = \mathbf{2.46 \times 10^{-3}} \textbf{ mol/L}](/tpl/images/0685/0322/0aa42.png)
![\text{pOH} = -\log \text{[OH}^{-}] = -\log(2.46 \times 10^{-3}) = 2.61](/tpl/images/0685/0322/acbc3.png)





