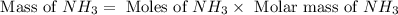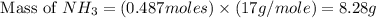Chemistry, 13.06.2020 14:57, snlawson9053
1.46 g H2 is allowed to react with 10.5 g N2, producing 2.72 g NH3. What is the theoretical yield in grams for this reaction under the given conditions? Express your answer to three significant figures and include the appropriate units.

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 10:30, tjjjjjjjjjjjjjjjjjjj
What determines the average kinetic energy of the particles in a gas? a. the number of collisions b. the number of particles c. the size of the particles d. the temperature
Answers: 1

Chemistry, 22.06.2019 17:00, emma3216
In a heat engine of 1000 j of heat enters the system and the piston does 500 j of work what is the final internal energy of the system if the inital energy was 2000 j we have to do all of these down here 1)write the equation 2)list out your know variables 3)plug the numbers into the equations 4)solve 5)write your solution statemtn that includes inital energuy and final energuy added
Answers: 1

Chemistry, 22.06.2019 22:30, arodavoarodavo
Which is a characteristic of the electron sea model for metallic bonding? molecular orbitals overlap to produce bands. electrons flow easily between metal nuclei. electrons are in fixed positions in the orbitals. atomic nuclei are arranged in an irregular pattern.
Answers: 3

Chemistry, 23.06.2019 03:50, KAITLYN007
Show how to convert the temperature 84.7° c to kelvin. include all steps and label the final answer.
Answers: 1
Do you know the correct answer?
1.46 g H2 is allowed to react with 10.5 g N2, producing 2.72 g NH3. What is the theoretical yield in...
Questions in other subjects:


Mathematics, 16.10.2019 01:10









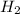 = 1.46 g
= 1.46 g
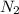 = 10.5 g
= 10.5 g
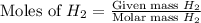
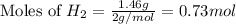
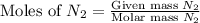
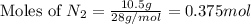
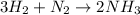
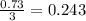 moles of
moles of 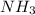
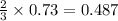 mole of
mole of