
Chemistry, 12.06.2020 15:57, carmencolon119
Given a K value of 0.43 for the following aqueous equilibrium, suppose sample Z is placed into water such that it’s original concentration is 0.033 M. Assume there was zero initial concentration of either A(aq) or B(aq). Once equilibrium has occurred, what will be the equilibrium concentration of Z? 2A(aq) + B(aq) <> 2Z (aq)

Answers: 3
Other questions on the subject: Chemistry



Chemistry, 22.06.2019 14:30, amylumey2005
How can carbon move from "land" to bodies of water? describe the way human impact has lead to increased levels of co2 in the atmosphere.
Answers: 2

Chemistry, 22.06.2019 18:00, heggestade
To apply in a gold the individual gold atoms are united to each other by means of a metallic bond. how would you use the gold block to determine the atomic radius of a gold atom?
Answers: 3
Do you know the correct answer?
Given a K value of 0.43 for the following aqueous equilibrium, suppose sample Z is placed into water...
Questions in other subjects:


Mathematics, 23.10.2020 21:50

Mathematics, 23.10.2020 21:50


Mathematics, 23.10.2020 21:50


Biology, 23.10.2020 21:50


English, 23.10.2020 21:50


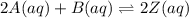
![K=\frac{[Z]^2}{[A]^2[B]}](/tpl/images/0684/0872/5d1cf.png)
![\frac{1}{K} =\frac{[A]^2[B]}{[Z]^2}=2.33](/tpl/images/0684/0872/c65f3.png)
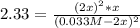
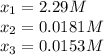
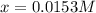 since the other solutions make the equilibrium concentration of Z negative which is not possible. In such a way, its concentration at equilibrium is:
since the other solutions make the equilibrium concentration of Z negative which is not possible. In such a way, its concentration at equilibrium is:![[Z]_{eq}=0.033M-2*0.0153M=0.0024M](/tpl/images/0684/0872/d1dc4.png)




