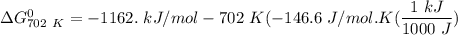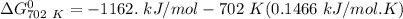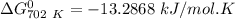Consider the following reaction:
2NO(g)+O2(g)→2NO2(g)
Estimate ΔG∘ for this reaction at each of the following temperatures and predict whether or not the reaction will be spontaneous. (Assume that ΔH∘ and ΔS∘ do not change too much within the give temperature range.) I need to find the temperature are 298K and 702K. For 298K It is simple because at standard temperature
ΔG∘ = DG(products)- DG(reactants).

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 10:00, alexabdercmur
Why is the structure of molecule important to its function?
Answers: 1

Chemistry, 22.06.2019 14:00, Killion2022
Anthracite is so hard and pure it is also referred to as a renewable resource metamorphic rock hot bituminous coal dirty fuel
Answers: 1

Chemistry, 22.06.2019 21:30, rondonalba
Electromagnets coils of wire paper clips picked up 10 3 15 6 20 9 25 12 ms. owens' class was studying magnets. ms. owens showed her students how to make an electromagnet using a nail, a d-cell battery, and plastic coated wire. the students wrapped the wire around the nail and then attached the ends to the battery. when they were finished, they tested their magnets by investigating how many paperclips their magnets could pick up. they also tested whether they could increase the strength of their electromagnets by using more coils of wire. they recorded the class average of their results in the data table seen here. ms. owens asked her students to graph their data in a line graph. how should the students label the x-axis on their line graph? a) size of battery b) number of paper clips c) number of coils of wire d) strength of electromagnet
Answers: 2

Chemistry, 23.06.2019 00:30, DragonLovely
•hydration •dissociation •dissolving which one goes to which
Answers: 1
Do you know the correct answer?
Consider the following reaction:
2NO(g)+O2(g)→2NO2(g)
Estimate ΔG∘ for this reaction at each...
Estimate ΔG∘ for this reaction at each...
Questions in other subjects:





Health, 28.06.2019 16:40






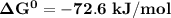 ; as such the reaction is said to be spontaneous since the value of
; as such the reaction is said to be spontaneous since the value of  is negative.
is negative. and the reaction is spontaneous
and the reaction is spontaneous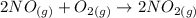
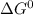 ;
;![\Delta G^0 = [2(\Delta G^0_{NO_{2(g)}}] - [1(\Delta G^0_{O_{2(g)}})+ 2(\Delta G^0_{NO_{g}})]](/tpl/images/0683/6875/905f6.png)
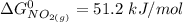
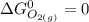
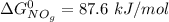
![\Delta G^0 = [2(51.2 \ kJ/mol}] - [1(0)+ 2(87.6 \ kJ/mol})]](/tpl/images/0683/6875/34022.png)
![\Delta G^0 = [102.4 \ kJ/mol}] - [175.2 \ kJ/mol})]](/tpl/images/0683/6875/a6424.png)
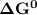 of the reaction when the temperature is 702 K is:
of the reaction when the temperature is 702 K is: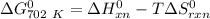
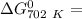 Gibbs free energy of the reaction at 702 K
Gibbs free energy of the reaction at 702 K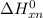 = standard enthalpy of the reaction = -116.2 kJ/mol
= standard enthalpy of the reaction = -116.2 kJ/mol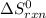 = standard entropy of the reaction = -146.6 J/mol/K
= standard entropy of the reaction = -146.6 J/mol/K