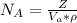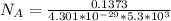Chemistry, 12.06.2020 00:57, cordobamariana07
3. Crystalline structural unit of barium metal is a body-centered cubic cell. The edge length of the unit cell is 5.02x10-8 cm. The density of the metal is 5.30 g/cm3. Assume that 68% of the unit cell is occupied by Ba atoms. The molar mass of barium is 137.3 g/mol. Using this information, calculate Avogadro’s number. Show your calculation procedure that allows you to derive Avogadro’s number. Your answer must show six digits after the decimal point (i. e., 6.pppx1023) that is not necessarily the same as the known value. By showing your calculation-result down to six digits after the decimal point, you showcase that you did calculate the number, instead of simply adopting the known Avogadro’s number available in open resources.

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 05:50, mrylenastewart
Fill in the coefficients that will balance the following reaction: a0cr2(so4)3 + a1agno3 -> a2cr(no3)3 + a3ag2so4
Answers: 1


Do you know the correct answer?
3. Crystalline structural unit of barium metal is a body-centered cubic cell. The edge length of the...
Questions in other subjects:






History, 10.05.2021 18:30

Spanish, 10.05.2021 18:30



English, 10.05.2021 18:30


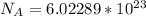
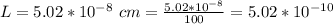
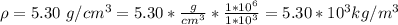
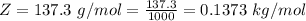
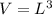
![V = [5.02 *10^{-10}]^3](/tpl/images/0683/5740/fad64.png)
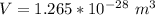
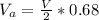
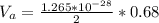
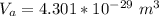
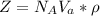
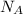 is the Avogadro's number
is the Avogadro's number