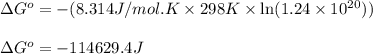In the activity, click on the Keq and ΔG∘ quantities to observe how they are related. Calculate ΔG∘using this relationship and the equilibrium constant (Keq) obtained in Part A at T=298K:Keq=1.24×1020Express the Gibbs free energy (ΔG∘) in joules to three significant figures.

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 23:00, ayoismeisalex
Matches the chemical name of each oxide of phosphorus to its chemical formula
Answers: 2

Chemistry, 22.06.2019 03:30, tbeck225
If a solution is considered basic, then a) the hydroxide ion and hydronium ion concentrations are equal. b) the hydroxide ion concentration is less than the hydronium ion concentration. c) the hydronium ion concentration is greater than the hydroxide ion concentration. d) the hydroxide ion concentration is greater than the hydronium ion concentration.
Answers: 1

Chemistry, 22.06.2019 12:30, fvmousdiana
In france, grapes are 1.95 euros per kilogram. what is the cost of grapes, in dollars per pound, if the exchange rate is 1.14 dollars/euro? (2.6)
Answers: 3
Do you know the correct answer?
In the activity, click on the Keq and ΔG∘ quantities to observe how they are related. Calculate ΔG∘u...
Questions in other subjects:


World Languages, 03.08.2019 19:30


English, 03.08.2019 19:30

Chemistry, 03.08.2019 19:30


Social Studies, 03.08.2019 19:30

Mathematics, 03.08.2019 19:30


Social Studies, 03.08.2019 19:30


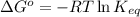
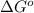 = Gibbs free energy of the reaction = ?
= Gibbs free energy of the reaction = ?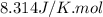
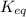 = equilibrium constant of the reaction =
= equilibrium constant of the reaction =