
Chemistry, 10.06.2020 06:57, smithsa10630
1.)A strong acid solution requires 3.2 grams of sulfuric acid (H2SO4). How many molecules of sulfuric acid are in the solution? 2.) While measuring out the sulfuric acid you accidentally spilled some of it! Before trying to clean it up you put some baking soda (NaHCO3) on to it neutralize it. If you scatter 7.8 g of baking soda on the acid how many moles of baking soda have you used?

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 23:00, brapmaster764
What is the formula that this ionic compounds could form sr2+p3-o2-
Answers: 3

Chemistry, 22.06.2019 04:00, lucasrandall
Asolution contains 225 g of sodium chloride, nacl, dissolved in enough water to make a 0.25 l of solution. what is the molarity of the solution?
Answers: 2


Chemistry, 22.06.2019 13:00, cnfndbxbfbdb2031
Which of the following are good traits of a hypothesis? it will be able to be testedit can predict an outcomeit will explain the observationsall of these
Answers: 2
Do you know the correct answer?
1.)A strong acid solution requires 3.2 grams of sulfuric acid (H2SO4). How many molecules of sulfuri...
Questions in other subjects:


Biology, 21.01.2022 14:00

Mathematics, 21.01.2022 14:00


Mathematics, 21.01.2022 14:00

Mathematics, 21.01.2022 14:00



Social Studies, 21.01.2022 14:00

History, 21.01.2022 14:00


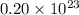 molecules of sulfuric acid in the solution.
molecules of sulfuric acid in the solution.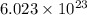 of particles.
of particles.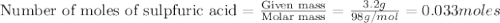
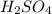 contains =
contains = 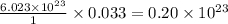 molecules of sulfuric acid
molecules of sulfuric acid 




