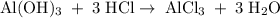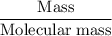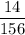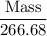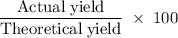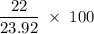Aluminum hydroxide is often present in antacids to neutralize stomach acid (HCl). If 14.0 g aluminum hydroxide is present in an antacid tablet, determine the theoretical yield of aluminum chloride produced when the tablet reacts with stomach acid. If the actual yield of the aluminum chloride from this tablet is 22.0 g, what is the percent yield?

Answers: 3
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 07:50, mckinleesmomp6qj1e
Which of the following electromagnetic waves can create ions?
Answers: 2

Chemistry, 22.06.2019 08:30, itzhari101
In a chemical reaction at equilibrium, the rate of the forward reaction the rate of the reverse reaction. if the rate of the forward reaction more products are formed.
Answers: 1

Do you know the correct answer?
Aluminum hydroxide is often present in antacids to neutralize stomach acid (HCl). If 14.0 g aluminum...
Questions in other subjects:


Business, 25.04.2020 22:48

Mathematics, 25.04.2020 22:48


Geography, 25.04.2020 22:48

Geography, 25.04.2020 22:48



Mathematics, 25.04.2020 22:48

Social Studies, 25.04.2020 22:48