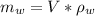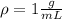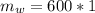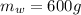Chemistry, 09.06.2020 23:57, jkw1222p0ttvq
A chemistry student is given 600. mL of a clear aqueous solution at 37° C. He is told an unknown amount of a certain compound X is dissolved in the solution. The student allows the solution to cool to 21° C. At that point, the student sees that a precipitate has formed. He pours off the remaining liquid solution, throws away the precipitate, and evaporates the water from the remaining liquid solution under vacuum. More precipitate forms. The student washes, dries and weighs the additional precipitate. It weighs 0.084 kg. Using only the information from above, can you calculate the solubility of X at 21° C?If yes, calculate it. Be sure your answer has a unit symbol and the right number of significant figures.

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 04:30, falishaduncanovmtz2
Electrons are extremely important to what area of technology? a) anti-aging research b) household product development c) electronics d) drug discovery
Answers: 3

Chemistry, 22.06.2019 08:20, pilarmonsivais
What is the formula for the compound dinitrogen pentoxide? a. n4o5 b. n5o4 c. n4o6 d. n5o2 e. n2o5
Answers: 3


Chemistry, 22.06.2019 14:30, Hannahmiller3773
Connect the whole numbers on the periodic table to indicate what they represent?
Answers: 3
Do you know the correct answer?
A chemistry student is given 600. mL of a clear aqueous solution at 37° C. He is told an unknown amo...
Questions in other subjects:


Mathematics, 19.02.2021 01:00

Mathematics, 19.02.2021 01:00


History, 19.02.2021 01:00



Mathematics, 19.02.2021 01:00

Mathematics, 19.02.2021 01:00

History, 19.02.2021 01:00


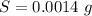
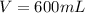
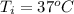
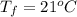
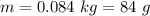
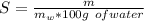
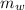 ) in the solution is mathematically represented as
) in the solution is mathematically represented as