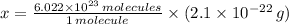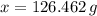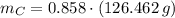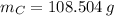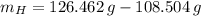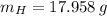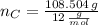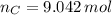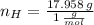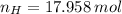Answers: 2
Other questions on the subject: Chemistry



Chemistry, 22.06.2019 15:00, emmalie52
Phosphorous acid, h3po3(aq) , is a diprotic oxyacid that is an important compound in industry and agriculture. p k a1 p k a2 1.30 6.70 calculate the ph for each of the points in the titration of 50.0 ml of 1.5 m h3po3(aq) 1.5 m h 3 po 3 ( aq ) with 1.5 m koh(aq). 1.5 m koh ( aq ) .
Answers: 1

Chemistry, 23.06.2019 02:00, Hellopeople233
Which of the following substances is the most soluble in water? a. sodium chloride b. methane c. bromine d. carbon
Answers: 1
Do you know the correct answer?
Un hidrocarburo tiene como composición en masa: C= 85.8% ; H= 14.2% . Como dato nos brindan que una...
Questions in other subjects:

Mathematics, 03.02.2020 01:54

History, 03.02.2020 01:54

Mathematics, 03.02.2020 01:54

Mathematics, 03.02.2020 01:54






History, 03.02.2020 01:54


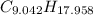
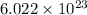 moléculas. La masa de un mol se determina mediante regla de tres simple:
moléculas. La masa de un mol se determina mediante regla de tres simple: