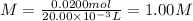Hydrobromic acid solution of unknown concentration is titrated with a 0.500M LiOH solution.
20.00mL of the acid are poured into an Erlenmeyer flask.
40.00mL of the base solution is required to reach the equivalence point.
What is the molarity of the Hydrobromic acid solution?

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 00:30, Bryanguzman2004
Water (2510 g ) is heated until it just begins to boil. if the water absorbs 5.09×105 j of heat in the process, what was the initial temperature of the water?
Answers: 3


Chemistry, 23.06.2019 02:00, bagofmud8339
The point along a planet's orbit where it is closest to the sun is called the
Answers: 1
Do you know the correct answer?
Hydrobromic acid solution of unknown concentration is titrated with a 0.500M LiOH solution.
20.00mL...
Questions in other subjects:


Physics, 01.04.2021 01:00




Mathematics, 01.04.2021 01:00

Physics, 01.04.2021 01:00