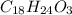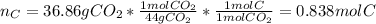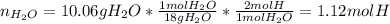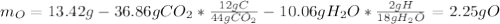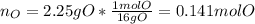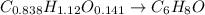Chemistry, 07.06.2020 05:00, calebwoodall6477
Combustion analysis of a 13.42-g sample of an unknown organic compound (which contains only carbon, hydrogen, and oxygen) produced 36.86 g CO2 and 10.06 g H2O. The molar mass of the compound is 288.38 g/mol . Part A Find the molecular formula of the unknown compound. Express your answer as a chemical formula.

Answers: 3
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 11:30, chelseychew32
What is the main reason why some developing countries fear the increase the free trade policies around the world?
Answers: 2


Chemistry, 22.06.2019 21:30, crystalbyrd79p8imrx
What is the effect of returning nuclear reactor cooling water back into bodies of water?
Answers: 3
Do you know the correct answer?
Combustion analysis of a 13.42-g sample of an unknown organic compound (which contains only carbon,...
Questions in other subjects:

Physics, 29.09.2021 23:40




History, 29.09.2021 23:40


Physics, 29.09.2021 23:40