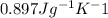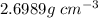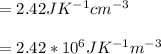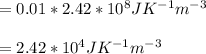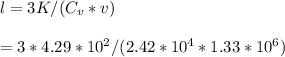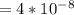Calculate the mean free path of electrons in a metal, such as silver, at room temperature form heat capacity and heat conduction measurements. Take EF ¼ 5 eV, K ¼ 4:29 102 J/s m K, and Cel v ¼ 1% of the lattice heat capacity. (Hint: Remember that the heat capacity in (21.8) is given per unit volume!)

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 16:00, anferneebcoleman
How many moles of oxygen react with 12 moles of aluminum
Answers: 1

Chemistry, 23.06.2019 07:20, prettydoll19
Which statement explains which component is likely to be more powerful in explaining a scientific phenomenon? a) component c, because a theory is often passed on possibility and not certainty b) component d, because a hypothesis is often based on possibility not certainty c) component c, because the ability to explain several occurrences in the natural world is a characteristic of a hypothesis d) component d, because the ability to explain several occurrences in the natural world is a characteristic of a theory
Answers: 3

Chemistry, 23.06.2019 08:30, vett072804
Benzonitrile (c6h5cn) is reduced to two different products depending on the reducing agent used. treatment with lithium aluminum hydride followed by water forms k, which has a molecular ion in its mass spectrum at 107 and the following ir absorptions: 3373, 3290, 3062, 2920, and 1600 cm-1. treatment with a milder reducing agent forms l, which has a molecular ion in its mass spectrum at 106 and the following ir absorptions: 3086, 2850, 2820, 2736, 1703, and 1600 cm-1. l shows fragments in its mass spectrum at m/z = 105 and 77. propose structures for k and l and choose an explanation for how this could be concluded.
Answers: 3
Do you know the correct answer?
Calculate the mean free path of electrons in a metal, such as silver, at room temperature form heat...
Questions in other subjects:


English, 08.07.2019 04:30

Mathematics, 08.07.2019 04:30


Business, 08.07.2019 04:30

Business, 08.07.2019 04:30


Biology, 08.07.2019 04:30



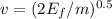
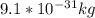 and
and 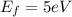 we get
we get 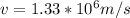
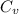 is 1% of lattice heat capacity
is 1% of lattice heat capacity