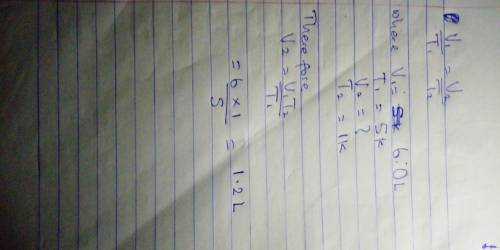Chemistry, 05.06.2020 06:00, kyleejokow
6.0 L of oxygen gas is at a temperature of 5K. If the temperature of the gas is lowered to 1K at constant pressure, what is the new volume of the gas?

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 21:00, bakoeboo
Harvey kept a balloon with a volume of 348 milliliters at 25.0˚c inside a freezer for a night. when he took it out, its new volume was 322 milliliters, but its pressure was the same. if the final temperature of the balloon is the same as the freezer’s, what is the temperature of the freezer? the temperature of the freezer is kelvins.
Answers: 2

Chemistry, 21.06.2019 22:00, NREYESLDS2806
To save time, you can approximate the initial mass of the solid to the nearest ±1 g. for example, if you are asked to add 14.3 g of copper, add between 13 g and 15 g. which of the following sets include two samples with an equal density? which all that apply below 15.4 g gold and 18.7 g silver 15.2 g copper and 50.0 g copper 20.2 g silver and 20.2 g copper 11.2 g gold and 14.9 g gold
Answers: 1

Chemistry, 22.06.2019 13:10, bartonamber4042
Select the correct answer a modure consists of glucose and water. what is the percent composition of glucose in the mixture if it contains 1.3 moles of glucose (cho total mass of the mature is 276 grams? ) and the a 1775
Answers: 1
Do you know the correct answer?
6.0 L of oxygen gas is at a temperature of 5K. If the temperature of the gas is lowered to 1K at con...
Questions in other subjects:


Mathematics, 22.01.2021 21:30

History, 22.01.2021 21:30

Spanish, 22.01.2021 21:30



English, 22.01.2021 21:30


Mathematics, 22.01.2021 21:30

Chemistry, 22.01.2021 21:30