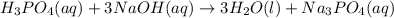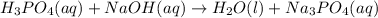Chemistry, 02.06.2020 23:00, angelinamadriga
In a neutralization reaction with a strong acid or base, all of the hydrogens from the acid and the hydroxides from the base react to form water.
Balance the following neutralization reaction:
H3PO4 (aq) + NaOH (aq) â H2O (l) + Na3PO4 (aq)
Enter the appropriate coefficient to the left of the respective term. In the event that your coefficient would be "1" be sure to write "1" as opposed to leaving it blank.

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 21:30, gatorr2010
An alcohol thermometer makes use of alcohol's changing in order to measure temperature. as the temperature goes up, the alcohol contained in the thermometer increases in volume, filling more of the thermometer's tube.
Answers: 3

Chemistry, 22.06.2019 08:00, wizz4865
Why is the bond angle in a water molecule less than the bond angle of methane? a. the central oxygen atom in water has two lone pairs of electrons, whereas the central carbon atom in methane has no lone pairs. b. the central hydrogen atom in water has one lone pair of electrons, whereas the central carbon atom in methane has two lone pairs. c. the central oxygen atom in water has four lone pairs of electrons, whereas the central carbon atom in methane has only one lone pair. d. the central oxygen atom exerts more repulsive force on surrounding atoms than the central carbon atom in methane does. reset next
Answers: 2

Chemistry, 22.06.2019 19:50, ellycleland16
Identify the lewis base in this balanced equation: fe3+ h2o fe(h2o)63+
Answers: 1

Do you know the correct answer?
In a neutralization reaction with a strong acid or base, all of the hydrogens from the acid and the...
Questions in other subjects:

Mathematics, 03.12.2020 23:20

Mathematics, 03.12.2020 23:20


History, 03.12.2020 23:20

Geography, 03.12.2020 23:20

Chemistry, 03.12.2020 23:20

Mathematics, 03.12.2020 23:20