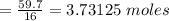Chemistry, 30.05.2020 00:03, 23jaywilliams
Given:
CH4 + 2O2 → CO2 + 2H2O, ΔH = -890 kJ/mol
How much energy is released when 59.7 grams of methane (CH4) reacts with oxygen?
The combustion of 59.7 grams of methane releases kilojoules of energy

Answers: 1
Other questions on the subject: Chemistry



Do you know the correct answer?
Given:
CH4 + 2O2 → CO2 + 2H2O, ΔH = -890 kJ/mol
How much energy is released when 5...
CH4 + 2O2 → CO2 + 2H2O, ΔH = -890 kJ/mol
How much energy is released when 5...
Questions in other subjects:

Mathematics, 15.10.2019 06:20


English, 15.10.2019 06:20


Biology, 15.10.2019 06:20



Mathematics, 15.10.2019 06:20


History, 15.10.2019 06:20