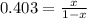Chemistry, 28.05.2020 04:57, jacquetpaul1969
A key step in the extraction of iron from its ore is FeO(s) + CO(g) ⇋ Fe(s) + CO2(g) Kp =0.403 at 1000°C. What are the equilibrium partial pressures of carbon monoxide and carbon dioxide when 1.00 atm of carbon monoxide and excess iron(II) oxide react in a sealed container at 1000°C. write Kp expression, do ICE chart with pressures, 0.287 atm CO2 and 0.713 atm CO

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 17:20, banna01man
Pegmatites are igneous rocks in which the individual minerals are very large. typically, the minerals are all light-colored quartz, feldspar and muscovite. if you were given a black and white photograph of a pegmatite in a quarry (where the rock has been blasted and broken), what physical properties could you use to identify those three minerals in this hypothetical photo? describe each mineral and the specific diagnostic properties. be specific.
Answers: 2

Chemistry, 22.06.2019 21:30, rileydavidharless
Which substance can be broken down by chemical means
Answers: 1


Chemistry, 23.06.2019 00:30, Keemdadream13
If there are 3.5 moles of koh, how many moles of naoh can be produced? question 1 options: a)3.0 moles naoh b)3.5 moles naoh c)1 moles naoh d)9 moles naoh
Answers: 1
Do you know the correct answer?
A key step in the extraction of iron from its ore is FeO(s) + CO(g) ⇋ Fe(s) + CO2(g) Kp =0.403 at 10...
Questions in other subjects:


Biology, 21.05.2021 04:20

History, 21.05.2021 04:20

English, 21.05.2021 04:20

Mathematics, 21.05.2021 04:20


Mathematics, 21.05.2021 04:20

Mathematics, 21.05.2021 04:20



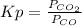 = 0.403
= 0.403