
Chemistry, 28.05.2020 01:01, floreschachi2873
5. A standardized mass was known to weigh exactly 5.0000g. It was weighed on a newly
acquired, uncalibrated balance as 5.001g. Calculate the absolute error and the percent error.

Answers: 3
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 12:00, daytonalive83481
Ican determine the molar mass of an element by looking on the under the atomic mass for the element. for example the molar mass of phosphorus is 30.974 grams/mole. avogadro’s number tells me the amount of representative particles in 1 mole of any substance. this means 12.011 gram sample of carbon and a 32.0 gram sample of sulfur have the same number of atoms.
Answers: 1

Chemistry, 22.06.2019 23:30, hcllxxhhlpcj
Rank substituents in order of their priority when assigning the e or z label to an alkene. i, ch2i , h, ch2ch2cl, f
Answers: 2

Chemistry, 23.06.2019 01:00, akluke6059
Which statement characterizes synthetic polymers? a. they come from animals and plants. b. they are found in nature. c. they are made in a lab. d. they are components of starch.
Answers: 1
Do you know the correct answer?
5. A standardized mass was known to weigh exactly 5.0000g. It was weighed on a newly
acquired,...
acquired,...
Questions in other subjects:


Mathematics, 02.02.2020 07:44

Biology, 02.02.2020 07:44


English, 02.02.2020 07:44

Mathematics, 02.02.2020 07:44

Mathematics, 02.02.2020 07:44



Mathematics, 02.02.2020 07:44


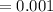
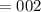 5
5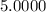 grams
grams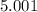 grams
grams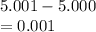
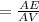
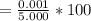
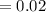 %
%



