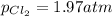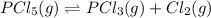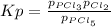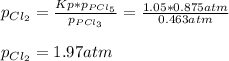Chemistry, 27.05.2020 23:06, hillarytrinh
3. The equilibrium constant KP for the decomposition of phosphorus pentachloride to phosphorus trichloride and molecular chlorine according to the chemical equation given below is found to be 1.05 at 250 °C. If the equilibrium partial pressures of PCl5 and PCl3 are 0.875 atm and 0.463 atm, respectively, what is the equilibrium partial pressure of Cl2 at 250 °C? PCl5(g) PCl3(g) + Cl2(g)

Answers: 2
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 11:30, charles8527
If blood contains 150g of hemoglobin per liter of blood, how much hemoglobin would be contained in 10 ml of blood
Answers: 2

Chemistry, 22.06.2019 19:30, Karinaccccc
How might this scientific phenomena be explained? a paper clip floats on water.
Answers: 1

Chemistry, 22.06.2019 21:50, SoccerAllStar2
Liquid from a brewery fermentation contains 10% ethanol and 90% water. part of the fermentation product (50,000 kg/h) is pumped to a distillation column on the factory site. under current operating conditions, a distillate of 45% ethanol and 55% water is produced from the top of the column at a rate of one-tenth that of the feed. what is the composition of the waste "bottoms" from the still?
Answers: 2
Do you know the correct answer?
3. The equilibrium constant KP for the decomposition of phosphorus pentachloride to phosphorus trich...
Questions in other subjects: