
Chemistry, 27.05.2020 08:58, destiny3200
C) Record the balanced chemical equation on the Student Worksheet.
Step 3: Determine the amount of energy change in the reaction.
a) Use the table of enthalpy values (Table A) provided in the Student Worksheet to locate the
enthalpy of formation (AH) for each reactant and each product. Record these values along with
the reactants and products in Table B of the Student Worksheet.
b) Determine the total enthalpy the reactants and the total enthalpy of the products. Record these
values in Table C of the Student Worksheet.
reactants)
c) Use the following formula to find the net change in enthalpy for the reaction and to determine
whether the reaction is endothermic or exothermic.
AH wen - (AH, products) -(AH
Record your answers in Table D.
Step 4: Model the energy change in the reaction.
a) Create an energy graph that illustrates the energy change in the reaction.
b) Construct your graph on a blank sheet of paper. Be sure to label the axes, provide a title, and
identify the reactants and products on the graph.
Step 5: Explain the energy change in the reaction.
a) Create a new blank document. Type your name at the top.
b) Write a few paragraphs describing the chemical reaction and explaining the energy change in
the reaction. Your document should
Did anyone do this project? And if so can you help me with these steps??

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 06:00, giusto1894
24. a sports ball is inflated to an internal pressure of 1.85 atm at room temperature (25 °c). if the ball is then played with outside where the temperature is 7.5 °c, what will be the new pressure of the ball? assume the ball does not change in volume nor does any air leak from the ball a) 0.555 atm b) 1.74 atm c) 1.85 atm d) 1.97 atm
Answers: 2



Do you know the correct answer?
C) Record the balanced chemical equation on the Student Worksheet.
Step 3: Determine the amoun...
Step 3: Determine the amoun...
Questions in other subjects:






Mathematics, 13.01.2021 18:00


Biology, 13.01.2021 18:00

Mathematics, 13.01.2021 18:00


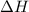 is positive then it means heat is absorbed by the system. And when
is positive then it means heat is absorbed by the system. And when 



