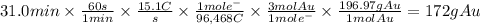Metal plating is done by passing a current through a metal solution. For example, an item can become gold plated by attaching the item to a power source and submerging it into an Au³⁺ solution. The item itself serves as the cathode, at which the Au³⁺ ions are reduced to Au(s). A piece of solid gold is used as the anode and is also connected to the power source, thus completing the circuit. What mass of gold is produced when 15.1 A of current are passed through a gold solution for 31.0 min?

Answers: 2
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 10:30, perezanthony2403
Which describes fat? a: a carbohydrate that produces energy b: a nucleic acid that directs cell function c: a lipid that stores energy d: a protein that speeds up a chemical reaction
Answers: 1

Chemistry, 22.06.2019 11:00, jodonw5616
The diagram below shows the different phase transitions that occur in matter. which arrow represents the transition in which dew is formed?
Answers: 1

Do you know the correct answer?
Metal plating is done by passing a current through a metal solution. For example, an item can become...
Questions in other subjects:


Social Studies, 26.01.2021 19:50

Biology, 26.01.2021 19:50

Mathematics, 26.01.2021 19:50

Mathematics, 26.01.2021 19:50



Mathematics, 26.01.2021 19:50

Mathematics, 26.01.2021 19:50

Mathematics, 26.01.2021 19:50