
Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 00:30, lasagnafoe
Used the balanced equation 2h2+ o2 - -> 2h2o. if you have 7.2 grams of o2 , how many grams of h2o can you produce ?
Answers: 2

Chemistry, 22.06.2019 01:30, MickeyxX7096
Phosphorous acid, h3po3(aq) , is a diprotic oxyacid that is an important compound in industry and agriculture. the values of phosphorous acid are 1.30 6.70 calculate the ph for each of the given points in the titration of 50.0 ml of 1.5 m h3po3(aq) with 1.5 m koh(aq) .
Answers: 3

Chemistry, 22.06.2019 06:00, tddreviews
Match the name of the following compound: mgso4 · h2omagnesium sulfate monohydratemagnesium (ii) sulfate monohydratemagnesium (ii) sulfate hydratemagnesium sulfate hydrate
Answers: 1
Do you know the correct answer?
How many atoms of Ca are in 4.7 moles of calcium?...
Questions in other subjects:

Biology, 23.07.2019 03:00


History, 23.07.2019 03:00

English, 23.07.2019 03:00

History, 23.07.2019 03:00

Mathematics, 23.07.2019 03:00

History, 23.07.2019 03:00

Health, 23.07.2019 03:00


Health, 23.07.2019 03:00


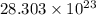 atoms in 4.7 moles of calcium.
atoms in 4.7 moles of calcium. 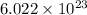 atoms.
atoms. 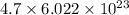 atoms
atoms



