
Chemistry, 22.05.2020 00:04, caromaybelline71
A first order reaction has rate constants of 4.6 x 10-2 s-1 and 8.1 x 10-2 s-1 at 0ºC and 20ºC, respectively. What is the value for the activation energy?
A.
0.566 J/mol
B.
2.5 x 10-4 J/mol
C.
2260 J/mol
D.
18,800 J/mol
E.
1.76 J/mol

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 22:30, pinkycupcakes3oxbqhx
200. ml of 3.00 m nacl solution is diluted to a final volume of 500. ml. what is the molarity of the final solution?
Answers: 2

Chemistry, 22.06.2019 14:30, Priskittles
What state of matter is ice a. liquid b. element c. solid d. gas
Answers: 1

Chemistry, 22.06.2019 18:00, AdoNice
Many pharmaceutical drugs are organic compounds that were originally synthesized in the laboratory. which two scientific disciplines are bridged by pharmaceutical drugs? chemistry and forensics chemistry and medicine biology and forensics biology and criminology
Answers: 2

Chemistry, 22.06.2019 22:50, kanerobertrosss2213
At the current rate, a graph of carbon dioxide produced by fossil fuels over time would slope upward slope downward be horizontal be vertical
Answers: 3
Do you know the correct answer?
A first order reaction has rate constants of 4.6 x 10-2 s-1 and 8.1 x 10-2 s-1 at 0ºC and 20ºC, resp...
Questions in other subjects:

World Languages, 30.10.2020 21:10

Biology, 30.10.2020 21:10

Mathematics, 30.10.2020 21:10

History, 30.10.2020 21:10

Mathematics, 30.10.2020 21:10

Mathematics, 30.10.2020 21:10


Biology, 30.10.2020 21:10

English, 30.10.2020 21:10


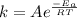 , where k is the rate constant, A is the frequency factor,
, where k is the rate constant, A is the frequency factor, 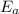 is the activation energy, R is the gas constant, and T is the temperature in Kelvins.
is the activation energy, R is the gas constant, and T is the temperature in Kelvins. . So:
. So: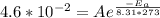
 . So:
. So:







