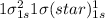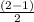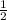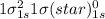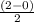Chemistry, 21.05.2020 05:10, korireidkdotdot82021
Use molecular orbital theory to determine whether He2 2+ or He2 + is more stable. Use molecular orbital theory to determine whether He2 2+ or He2 + is more stable. The He2 + ion is more stable since it has a higher bond order (bond order = 1) than the He2 2+ ion (bond order = 1/2). The He2 + ion is more stable since it has a higher bond order (bond order = 2) than the He2 2+ ion (bond order = 1). The He2 2+ ion is more stable since it has a lower bond order (bond order = 1/2) than the He2 + ion (bond order = 1). The He2 2+ a lower bond order (bond order = 3/2) than the He2 + ion (bond order = 2). The He2 2+ ion is more stable since it has a higher bond order (bond order = 1) than the He2 + ion (bond order = 1/2).

Answers: 2
Other questions on the subject: Chemistry


Chemistry, 21.06.2019 22:30, kkruvc
Will mark brainliest 26. which of these statements are true? (3 points) a. gases are compressible b. gases fill their containers completely c. the pressure of a gas is independent of the temperature d. gases have mass e. gases exert pressure f. the pressure of a gas is dependent on the volume g. gas pressure results from the collisions between gas particles h. gases have a definite volume and shape
Answers: 1

Chemistry, 22.06.2019 04:30, homeschool0123
How many moles of air are there in a human lung with a volume of 2.4 l at stp? explain your answer
Answers: 1

Chemistry, 22.06.2019 04:40, marknjenbennetp3j1v1
Listen base your answer to the question on the information below. propane is a fuel that is sold in rigid, pressurized cylinders. most of the propane in a cylinder is liquid, with gas in the space above the liquid level. when propane is released from the cylinder, the propane leaves the cylinder as a gas. propane gas is used as a fuel by mixing it with oxygen in the air and igniting the mixture, as represented by the balanced equation below. c3h8(g) + 5o2(g) → 3co2(g) + 4h2o() + 2219.2 kja small amount of methanethiol, which has a distinct odor, is added to the propane to consumers detect a propane leak. in methanethiol, the odor is caused by the thiol functional group (–sh). methanethiol, ch3sh, has a structure that is very similar to the structure of methanol. what is the correct structural formula for a molecule of methanethiol
Answers: 3
Do you know the correct answer?
Use molecular orbital theory to determine whether He2 2+ or He2 + is more stable. Use molecular orbi...
Questions in other subjects:

Mathematics, 02.12.2021 16:20


Chemistry, 02.12.2021 16:20


English, 02.12.2021 16:20

Engineering, 02.12.2021 16:20

Mathematics, 02.12.2021 16:20

English, 02.12.2021 16:20

Mathematics, 02.12.2021 16:20

Mathematics, 02.12.2021 16:20