Chemistry, 19.05.2020 03:09, tejasheree
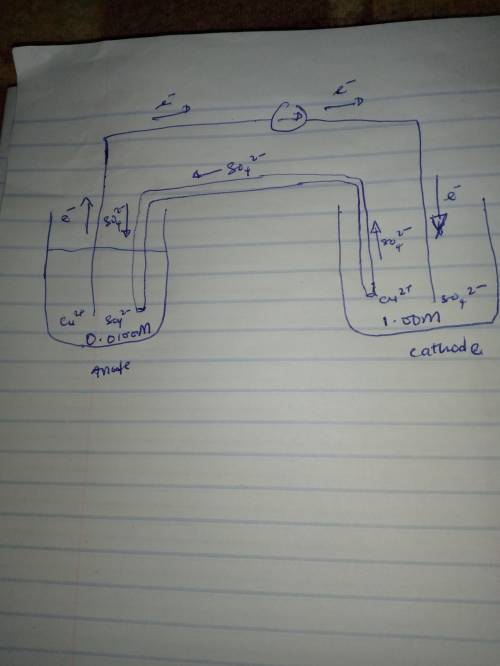
) A concentration cell is based on the aqueous reaction
Cu2+(1.00 M) Cu2+(0.0100 M)
The cell consists of copper electrodes dipping into solutions of Cu2+ ions. The anions present are sulfate ions. Draw a neat diagram to represent this cell, showing and labeling all necessary components including: anode, cathode, electron flow, cation flow and anion flow.

Answers: 1
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 14:30, clemsongirl5392
Select all that apply. using a value of ksp = 1.8 x 10-2 for the reaction pbcl2 (s) pb+2(aq) + 2cl -(aq). the concentration of the products yield a ksp of 2.1 x 10-2:
Answers: 2


Chemistry, 22.06.2019 18:40, johnnysteeler9934
What is one real world example of a colligative property?
Answers: 2
Do you know the correct answer?
) A concentration cell is based on the aqueous reaction
Cu2+(1.00 M) Cu2+(0.0100 M)
Th...
Cu2+(1.00 M) Cu2+(0.0100 M)
Th...
Questions in other subjects:

Business, 02.07.2021 15:20


Chemistry, 02.07.2021 15:20