
Chemistry, 13.05.2020 09:57, hannahrasco4051
The activation energy for proline isomerization of a peptide depends on the identity of the preceding residue and obeys Arrhenius rate behavior. Experiments are conducted on the isomerization of an alanine- proline peptide. At 25°C (298 K) the observed rate constant is 0.05 sec–1 and the value of EA is calculated to be 60 kJ•mol–1. Similar measurements are performed on a phenylalanine-proline peptide at 25°C, with a measured rate constant of 0.005 sec–1. Assuming an identical preexponential factor as the alanine-proline peptide, what is the activation energy for this peptide (kJ/mol)?

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 00:30, rscott2649
In numbering carbon atoms in the parent chain of a hydrocarbon, why would you number from right to left, rather than left to right
Answers: 1

Chemistry, 22.06.2019 09:20, lanaiheart7
How have the greenhouse gasses increased from the year 2000 to 2018
Answers: 2


Do you know the correct answer?
The activation energy for proline isomerization of a peptide depends on the identity of the precedin...
Questions in other subjects:

Mathematics, 03.12.2020 06:30


Mathematics, 03.12.2020 06:30


Chemistry, 03.12.2020 06:30

Social Studies, 03.12.2020 06:30



Mathematics, 03.12.2020 06:30

English, 03.12.2020 06:30


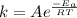 , where k is rate constant, A is pre-exponential factor,
, where k is rate constant, A is pre-exponential factor, 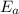 is activation energy, R is gas constant and T is temperature in kelvin scale.
is activation energy, R is gas constant and T is temperature in kelvin scale.![\frac{k_{ala-pro}}{k_{phe-pro}}=e^\frac{[E_{a}^{phe-pro}-E_{a}^{ala-pro}]}{RT}](/tpl/images/0653/2427/f6d0c.png)
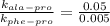 , T = 298 K , R = 8.314 J/(mol.K) and
, T = 298 K , R = 8.314 J/(mol.K) and 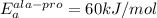
![\frac{0.05}{0.005}=e^{\frac{[E_{a}^{phe-pro}-(60000J/mol)]}{8.314J.mol^{-1}.K^{-1}\times 298K}}](/tpl/images/0653/2427/26a26.png)

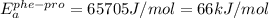 (rounded off to two significant digit)
(rounded off to two significant digit)



