
Chemistry, 07.05.2020 02:04, Svetakotok
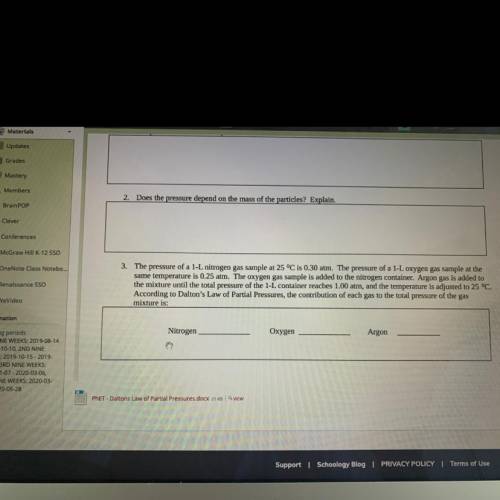
The pressure of a 1-L nitrogen gas sample at 25 ⁰C is 0.30 atm. The pressure of a 1-L oxygen gas sample at the same temperature is 0.25 atm. The oxygen gas sample is added to the nitrogen container. Argon gas is added to the mixture until the total pressure of the 1-L container reaches 1.00 atm, and the temperature is adjusted to 25 ⁰C. According to Dalton’s Law of Partial Pressures, the contribution of each gas to the total pressure of the gas mixture is:

Answers: 3
Other questions on the subject: Chemistry



Chemistry, 22.06.2019 09:00, tashaunalewis4786
This chart lists four kinds of polymers and their sources. what can be known about all four polymers, despite their differences? they come from living things. they share ionic carbon bonds. they are at least 100 monomers long. they are made of repeating subunits.
Answers: 1
Do you know the correct answer?
The pressure of a 1-L nitrogen gas sample at 25 ⁰C is 0.30 atm. The pressure of a 1-L oxygen gas sam...
Questions in other subjects:

Mathematics, 29.07.2019 08:30

Mathematics, 29.07.2019 08:30

Mathematics, 29.07.2019 08:30


Business, 29.07.2019 08:30

History, 29.07.2019 08:30

Chemistry, 29.07.2019 08:30



Mathematics, 29.07.2019 08:30






