
Sodium (Na) reacts with nitrogen gas (N2) to produce sodium nitride. If a student collects 2.5 moles of sodium nitride (NaN3) after the reaction has been completed, determine the number of moles of nitrogen that were initially present.
2Na + 3N2 -> 2NaN3
Your
3.75 mole N2
1.2 mole N2
3.87 mole N2

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 10:30, kdenormandie3122
Geothermal energy for industrial use is available almost anywhere. a. true b. false
Answers: 2


Chemistry, 22.06.2019 19:40, powberier6979
What is the wavelength of a 3*10^12 hz infrared wave a 3*10^20m b 1* 10^4m c 3*10^-3m d 1*10^-4 m
Answers: 1

Chemistry, 23.06.2019 00:00, vanessacox45
Total the mass on the syringe. record it in the correct row of the data table. kg done click and drag weights to change the pressure. click the syringe to zoom in and see the volume. intro
Answers: 3
Do you know the correct answer?
Sodium (Na) reacts with nitrogen gas (N2) to produce sodium nitride. If a student collects 2.5 moles...
Questions in other subjects:


History, 30.10.2020 06:10




Mathematics, 30.10.2020 06:10

Chemistry, 30.10.2020 06:10


English, 30.10.2020 06:10

Mathematics, 30.10.2020 06:10


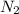
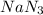 = 2.5 mol
= 2.5 mol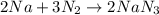
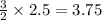 moles of
moles of 



