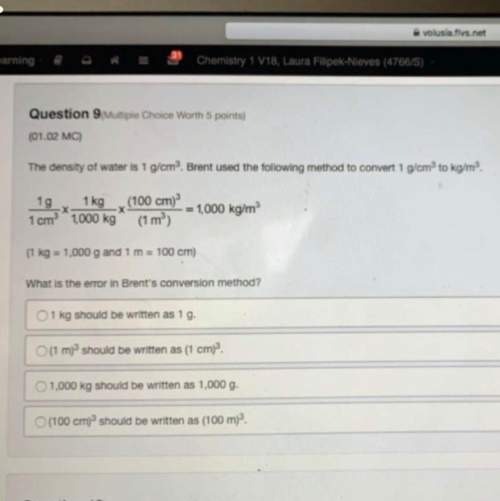
The chlorination of the iron (III) ion is an endothermic reaction.
Fe^3+(aq) + 2Cl-(aq)...

The chlorination of the iron (III) ion is an endothermic reaction.
Fe^3+(aq) + 2Cl-(aq) ↔ FeCl^2+(aq)
Predict the effect of increasing the reaction temperature.
A) no effect
B) increase in Cl-(aq) concentration
C) increase in Fe3+(aq) concentration
D) increase in FeCl2+(aq) concentration

Answers: 3
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 00:30, catdog5225
Drive down any three characteristic of modern periodic table
Answers: 1

Chemistry, 22.06.2019 06:00, palomaresmitchelle
There are 6.022, 104 atoms of hg in 1 mole of hg the number of atoms in 45 moles of hg can be found by multiplying 4.5 by 6.022, 102 which is the number of atoms in 4.5 moles of hg, correctly written in scientific notation with the correct number of significant figures? 0 21,109 0 21,100 271, 1024 27.099, 100 mark this and retum save and exit submit
Answers: 1

Chemistry, 22.06.2019 17:30, kevin72937
Consider the story you just read. all but one of the choices below indicate that something is living.
Answers: 1
Do you know the correct answer?
Questions in other subjects:

Biology, 19.01.2020 23:31

Social Studies, 19.01.2020 23:31




Mathematics, 19.01.2020 23:31