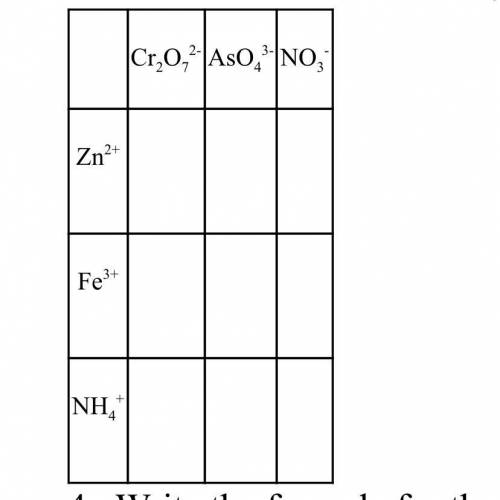
The answers to fill in this table
...

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 02:50, JuniperGalaxy
Consider the equilibrium system: 2icl(s) ⇄ i2(s) + cl2(g) which of the following changes will increase the total amount of of cl2 that can be produced? all of the listed answers are correct decreasing the volume of the container removing the cl2 as it is formed adding more icl(s) removing some of the i2(s)
Answers: 1

Chemistry, 22.06.2019 04:30, logan12345677885675
What are the primary responsibilities of a chemical engineer involved in "r& d"? develop large scale manufacturing operations discover new products and processes training of new chemists determine products needed by consumers
Answers: 2

Do you know the correct answer?
Questions in other subjects:

Mathematics, 08.12.2020 18:40

Mathematics, 08.12.2020 18:40

Mathematics, 08.12.2020 18:40



Mathematics, 08.12.2020 18:40


Mathematics, 08.12.2020 18:40


Advanced Placement (AP), 08.12.2020 18:40