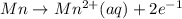A galvanic cell consists of one half-cell that contains Ag(s) and Ag+(aq), and one half-cell that contains Mn(s) and Mn2+(aq). What species are produced at the electrodes under standard conditions? Ag+(aq) + e- → Ag(s) E° = +0.80 V Mn2+(aq) + 2 e- → Mn(s) E° = -1.18 V A) Ag(aq) is formed at the cathode and, Mn(s) is formed at the anode. B) Ag(s) is formed at the cathode, and Mn2+(aq) is formed at the anode. C) Mn(s) is formed at the cathode, and Ag+(aq) is formed at the anode. D) Mn2+(aq) is formed at the cathode, and Ag(s) is formed at the anode.

Answers: 3
Other questions on the subject: Chemistry



Do you know the correct answer?
A galvanic cell consists of one half-cell that contains Ag(s) and Ag+(aq), and one half-cell that co...
Questions in other subjects:


Mathematics, 29.05.2020 04:03


Mathematics, 29.05.2020 04:03

Chemistry, 29.05.2020 04:03


English, 29.05.2020 04:03

Mathematics, 29.05.2020 04:03

Physics, 29.05.2020 04:03


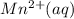 is formed at the anode.
is formed at the anode. 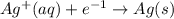 E=0.80 V
E=0.80 V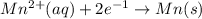 E=-1.18 V
E=-1.18 V