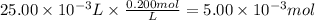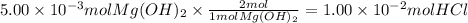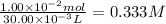Chemistry, 06.05.2020 01:39, kell22wolf
What is the molarity of a hydrochloric acid solution, HCl (aq), if 30.00 mL of the solution is required to completely react with 25.00 mL of a 0.200 M magnesium hydroxide solution, Mg(OH)2 (aq)? Be sure to write out the balanced chemical
reaction between these two compounds.

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 01:20, whrjegt4jrnfdvj
1. suppose a reaction mixture, when diluted with water, afforded 300 ml of an aqueous solution of 30 g of the reaction product malononitrile [ch2(cn)2], which is to be isolated by extraction with ether. the solubility of malononitrile in ether at room temperature is 20.0 g/100 ml, and in water is 13.3 g/100 ml. what weight of malononitrile would be recovered by extraction with (a) three 100-ml portions of ether and (b) one 300-ml portion of ether? suggestion: for each extraction, let x equal the weight extracted into the ether layer. in part (a), the concentration in the ether layer is x/100 and in the water layer is (30 x)/300; the ratio of these quantities is equal to k 20/13.3.
Answers: 2

Chemistry, 22.06.2019 10:10, jojomgarcia01
When water dissociates, each water molecule splits into a hydroxide ion and a) h 3 o + b) a hydrogen atom c) a hydrogen ion d) h 2 o e) oh —
Answers: 2


Chemistry, 23.06.2019 01:00, breemills9552
What two factors can affect the properties of a hydrocarbon? a. the number of its carbon atoms and the number of single bonds b. the number of its carbon atoms and the arrangement of its atoms c. the arrangement of its atoms and the number of its double bonds
Answers: 1
Do you know the correct answer?
What is the molarity of a hydrochloric acid solution, HCl (aq), if 30.00 mL of the solution is requi...
Questions in other subjects:

English, 18.09.2021 06:30




Physics, 18.09.2021 06:30

Mathematics, 18.09.2021 06:30

Mathematics, 18.09.2021 06:30

Mathematics, 18.09.2021 06:30

Mathematics, 18.09.2021 06:30

Mathematics, 18.09.2021 06:30