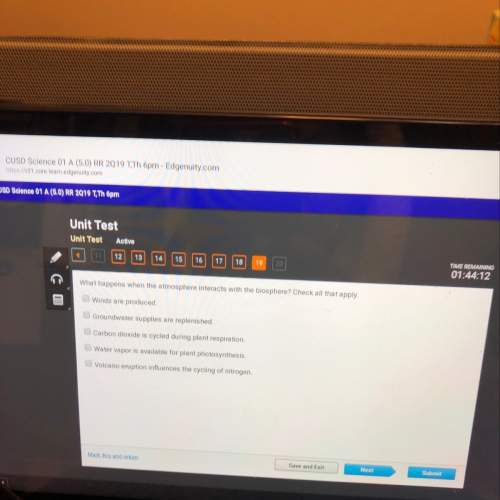
A scientist takes a small, partially inflated balloon
out of liquid nitrogen (at a very low te...

A scientist takes a small, partially inflated balloon
out of liquid nitrogen (at a very low temperature).
As the balloon rests on the table, it begins to grow
in size. Which statement(s) best explains why the
balloon grows?
The air from the outside comes inside the
balloon. More air means more volume.
The air warms and increases particle
movement. Increased particle movement
leads to increased volume.
side
o
Increased temperature means increased
volume as long as the container is flexible.

Answers: 2
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 09:40, kolibeilfuss
Sulfur dioxide and oxygen react to form sulfur trioxide during one of the key steps in sulfuric acid synthesis. an industrial chemist studying this reaction fills a 25.0l tank with 4.5 mol of sulfur dioxide gas and 4.5 mol of oxygen gas at 30.°c. he then raises the temperature, and when the mixture has come to equilibrium measures the amount of sulfur trioxide gas to be 1.4 mol. calculate the concentration equilibrium constant for the reaction of sulfur dioxide and oxygen at the final temperature of the mixture. round your answer to 2 significant digits.
Answers: 3


Do you know the correct answer?
Questions in other subjects: