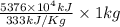On a hot summer day you and some friends decide you want to cool down your pool. Determine the mass of ice you would need to add to bring the equilibrium temperature of the system to 300K. The pool contains 400,000 L (at a density of 1 kg/L) of water initially at 305K. Assume the ice is at 0°C (273K), the heat capacity of water is 4.2 J/(g*K), and the enthalpy of melting ice is 333 kJ/kg.

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 19:30, itzyagirlshy
Determine the number o moles of ions/atoms/particle in the following: 2.50 miles of k2s (let me know how to do)
Answers: 1



Chemistry, 22.06.2019 06:30, backup5485
Three cards with holes are arranged in a straight line. a light is shined through the first card’s hole and travels through all three cards. what does this tell you about light rays? a) that light is reflected b) that light is refractive c) that light travels in a straight line d) that light does not travel in a straight line
Answers: 1
Do you know the correct answer?
On a hot summer day you and some friends decide you want to cool down your pool. Determine the mass...
Questions in other subjects:





Mathematics, 09.11.2020 03:40






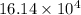 kg.
kg.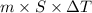
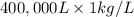
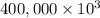 g (as 1 kg = 1000 g)
g (as 1 kg = 1000 g) = 305 K - 273 K
= 305 K - 273 K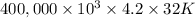
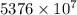 J
J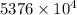 kJ
kJ