Chemistry, 05.05.2020 14:56, phillipfruge3
What is the freezing point of a 0.82 m aqueous solution of a non-volatile non-electrolyte?
PLEASE HELP

Answers: 3
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 16:00, anferneebcoleman
How many moles of oxygen react with 12 moles of aluminum
Answers: 1

Chemistry, 22.06.2019 20:20, carcon2019
Nitric acid can be formed in two steps from the atmospheric gases nitrogen and oxygen, plus hydrogen prepared by reforming natural gas. in the first step, nitrogen and hydrogen react to form ammonia: (g) (g) (g) in the second step, ammonia and oxygen react to form nitric acid and water: (g) (g) (g) (g) calculate the net change in enthalpy for the formation of one mole of nitric acid from nitrogen, hydrogen and oxygen from these reactions. round your answer to the nearest .
Answers: 3

Chemistry, 23.06.2019 07:30, fernandancon1872
Assume that 13.5 g solid aluminum (al) react with hcl to produce solid aluminum chloride (alcl3) salt and gaseous hydrogen (h2) at standard temperature and pressure.
Answers: 1
Do you know the correct answer?
What is the freezing point of a 0.82 m aqueous solution of a non-volatile non-electrolyte?
Questions in other subjects:


Biology, 07.12.2019 21:31

Social Studies, 07.12.2019 21:31

Mathematics, 07.12.2019 21:31

Computers and Technology, 07.12.2019 21:31


Mathematics, 07.12.2019 21:31



Chemistry, 07.12.2019 21:31


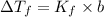
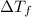 : the freezing-point depression
: the freezing-point depression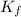 : cryoscopic constant (For water, Kf = 1.86 °C/m)
: cryoscopic constant (For water, Kf = 1.86 °C/m)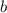 : molality
: molality