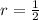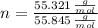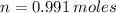Chemistry, 05.05.2020 00:28, bricksaspares
Iron and oxygen form rust. How many moles of rust should be produced if 1
55.321 grams of iron reacted? *

Answers: 1
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 14:50, jonmorton159
Consider the following multistep reaction: a b→ab(slow) a ab→a2b(fast)−−−−−−−−−−−−−−−−− 2a b→a2b(overall) based on this mechanism, determine the rate law for the overall reaction. express your answer in standard masteringchemistry format. for example, if the rate law is k[a]3[b]2 type k*[a]^3*[b]^2
Answers: 3

Chemistry, 22.06.2019 15:00, MilanPatel
How is the shape of the poem “peer” connected to its meaning?
Answers: 2

Chemistry, 22.06.2019 18:00, ambarpena14
An object displaces 652 ml of water. the volume of the object is: 0.652 cm³ 6.52 cm³ 65.2 cm³ 652 cm³
Answers: 2
Do you know the correct answer?
Iron and oxygen form rust. How many moles of rust should be produced if 1
55.321 grams of iron...
55.321 grams of iron...
Questions in other subjects:

Mathematics, 20.09.2020 03:01

Mathematics, 20.09.2020 03:01


Mathematics, 20.09.2020 03:01


English, 20.09.2020 03:01



Mathematics, 20.09.2020 03:01