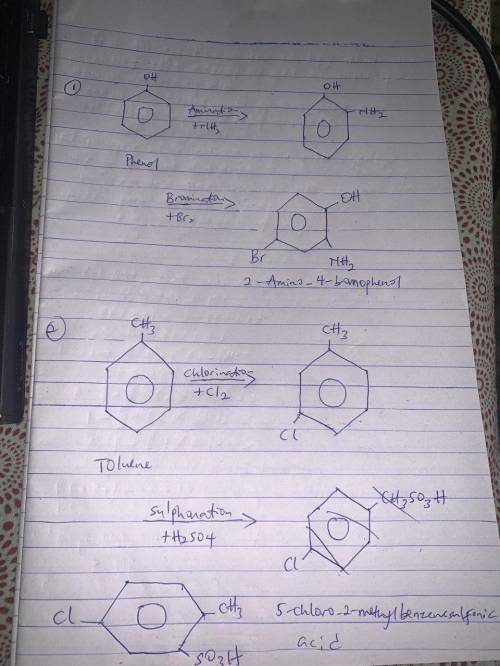
Show how you would synthesize each compound from benzene, toluene, or phenol using the following reactions: Choose the starting material from the drop-down list Enter the number(s) of the desired reactions in the order that you wish to use them without commas between If a reaction is used more than once, enter its number each time you wish to use it Compound 1: 2-amino-4-bromophenol Starting material: Reaction(s): Compound 2: 5-chloro-2-methylbenzenesulfonic acid Starting material: Reaction(s):

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 00:30, TMeansStupidity
Jessica is traveling from miami, florida, to chicago, illinois. using the map, tell one way the land will change during the second half of her trip.
Answers: 1

Chemistry, 22.06.2019 16:00, corcoranrobert1959
How do dying stars contribute to the formation of planets
Answers: 1

Chemistry, 22.06.2019 21:20, skyemichellec
40dm3 of gas at 760 torr are heated from 5°c to 50°c what is the new volume
Answers: 3
Do you know the correct answer?
Show how you would synthesize each compound from benzene, toluene, or phenol using the following rea...
Questions in other subjects:

English, 26.08.2019 05:30

Social Studies, 26.08.2019 05:30


Biology, 26.08.2019 05:30


Computers and Technology, 26.08.2019 05:30