Chemistry, 05.05.2020 04:06, 20alondra04
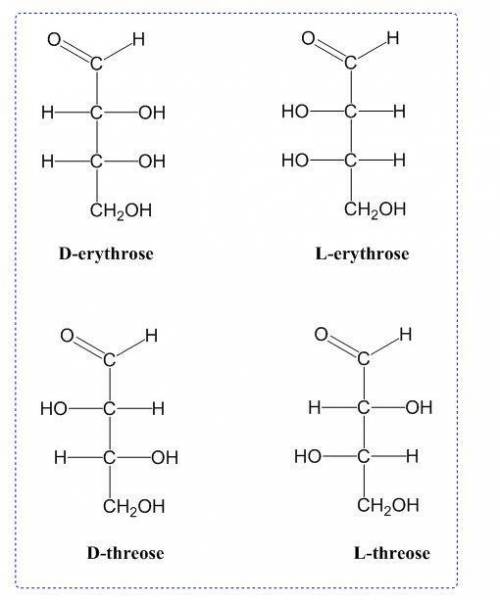
Draw the Fischer projections of the four aldotetroses. Draw the D sugar on the left and its L isomer directly to the right of it. Switch between an aldose and a ketose by clicking on \"switch carbonyl group.\" Add or delete carbon atoms using the add (\" \") or delete (\"x\") buttons. Clicking on a blue box once adds a hydrogen atom (H). Clicking on a blue box again toggles between H and OH.

Answers: 3
Other questions on the subject: Chemistry



Chemistry, 22.06.2019 12:00, Unknowndragon42
Consider the following reaction at equilibrium. 2co2 (g) 2co (g) + o2 (g) h° = -514 kj le châtelier's principle predicts that the equilibrium partial pressure of co (g) can be maximized by carrying out the reaction a. at high temperature and high pressure b. at high temperature and low pressure c. at low temperature and low pressure d. at low temperature and high pressure e. in the presence of solid carbon
Answers: 2

Chemistry, 22.06.2019 14:50, rebeccamckellpidge
The table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 9 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of tthe table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 9 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of the two atoms? both are unreactive. both are highly reactive. a is unreactive and d is reactive. a is reactive and d is unreactive.
Answers: 3
Do you know the correct answer?
Draw the Fischer projections of the four aldotetroses. Draw the D sugar on the left and its L isomer...
Questions in other subjects:

Computers and Technology, 14.09.2020 16:01

English, 14.09.2020 16:01

Mathematics, 14.09.2020 16:01

Mathematics, 14.09.2020 16:01

Mathematics, 14.09.2020 16:01

Mathematics, 14.09.2020 16:01

Geography, 14.09.2020 16:01

Mathematics, 14.09.2020 16:01

Biology, 14.09.2020 16:01

Mathematics, 14.09.2020 16:01