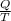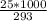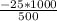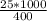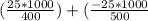Chemistry, 05.05.2020 05:40, olejlund8073
An object at 20∘C absorbs 25.0 J of heat. What is the change in entropy ΔS of the object? Express your answer numerically in joules per kelvin. ΔS = nothing J/K Request Answer Part E An object at 500 K dissipates 25.0 kJ of heat into the surroundings. What is the change in entropy ΔS of the object? Assume that the temperature of the object does not change appreciably in the process. Express your answer numerically in joules per kelvin. ΔS = nothing J/K Request Answer Part F An object at 400 K absorbs 25.0 kJ of heat from the surroundings. What is the change in entropy ΔS of the object? Assume that the temperature of the object does not change appreciably in the process. Express your answer numerically in joules per kelvin. ΔS = nothing J/K Request Answer Part G Two objects form a closed system. One object, which is at 400 K, absorbs 25.0 kJ of heat from the other object, which is at 500 K. What is the net change in entropy ΔSsys of the system? Assume that the temperatures of the objects do not change appreciably in the process. Express your answer numerically in joules per kelvin.

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 00:00, melidacampos12
Substance x has a fixed volume, and the attraction between its particles is strong .substance y had widely spread out particles and can be compressed what can most likely be concluded about these substances
Answers: 2



Do you know the correct answer?
An object at 20∘C absorbs 25.0 J of heat. What is the change in entropy ΔS of the object? Express yo...
Questions in other subjects:

Mathematics, 05.10.2021 06:50

Engineering, 05.10.2021 06:50

Mathematics, 05.10.2021 06:50

Computers and Technology, 05.10.2021 06:50

Chemistry, 05.10.2021 06:50

Business, 05.10.2021 06:50

Physics, 05.10.2021 06:50

Geography, 05.10.2021 06:50

Mathematics, 05.10.2021 06:50

English, 05.10.2021 06:50